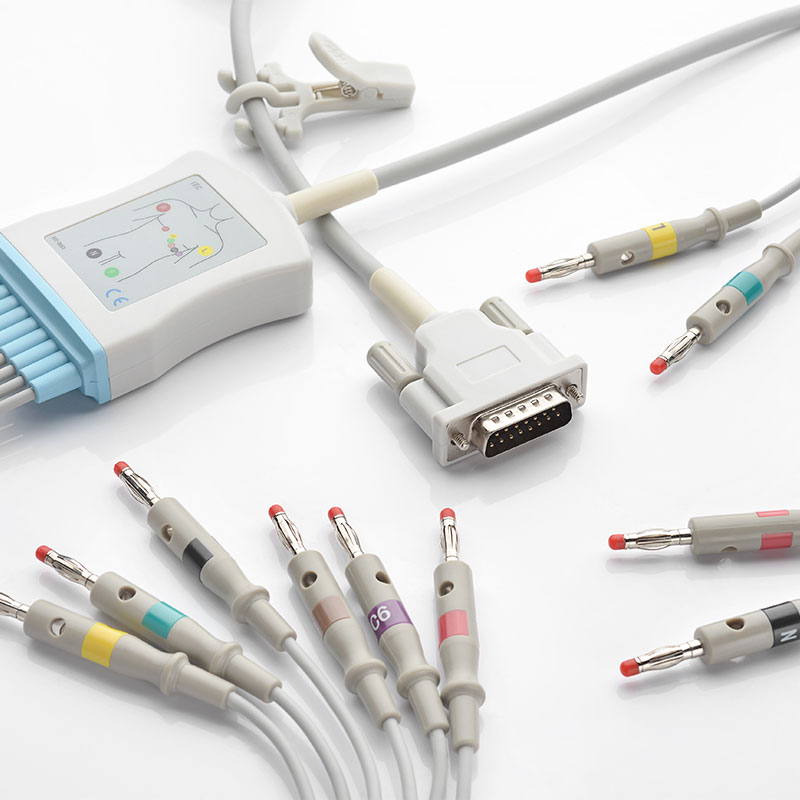
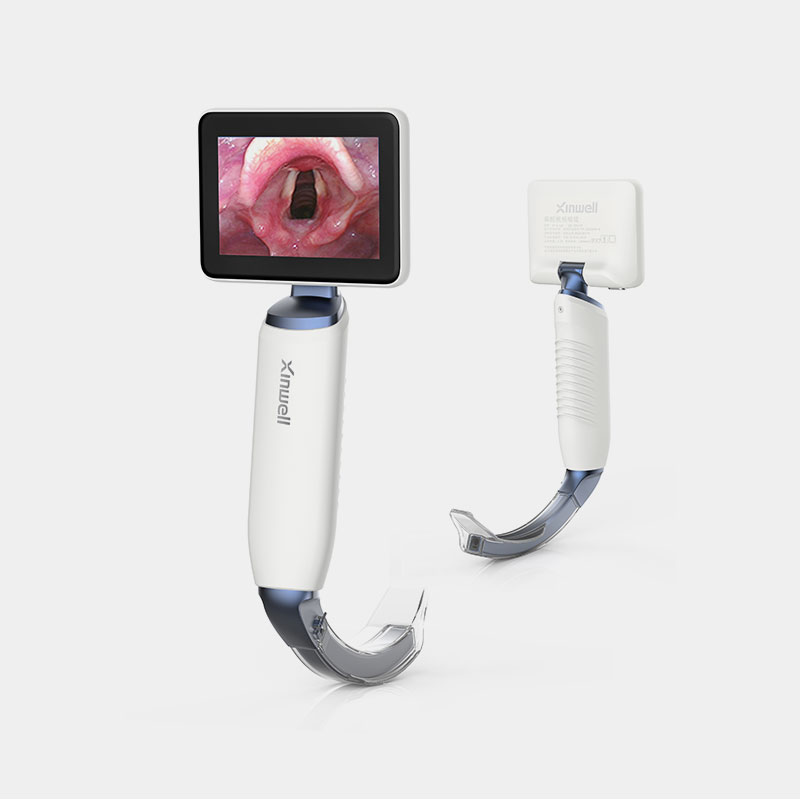
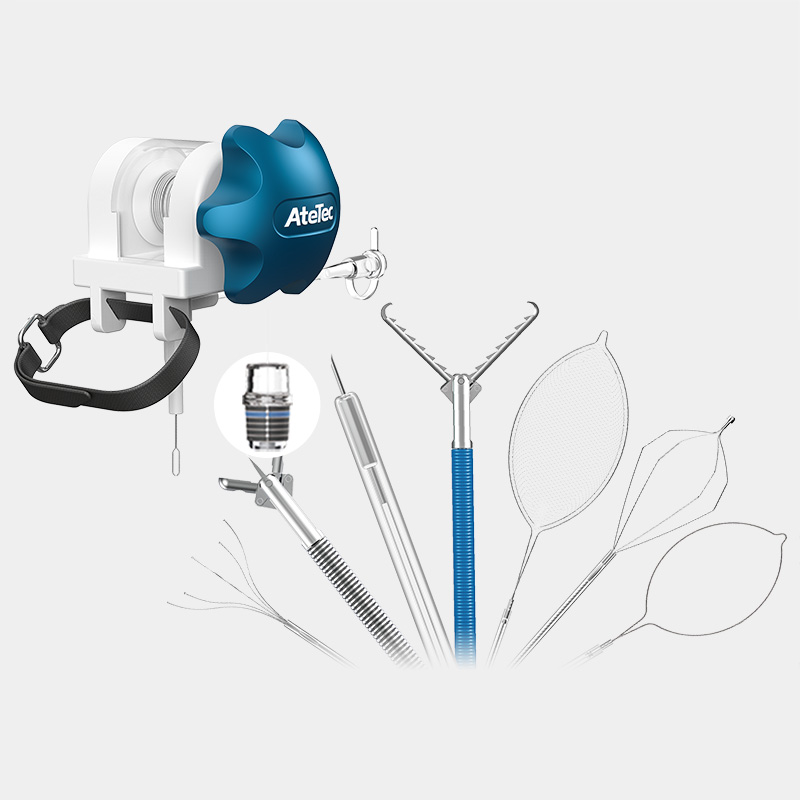
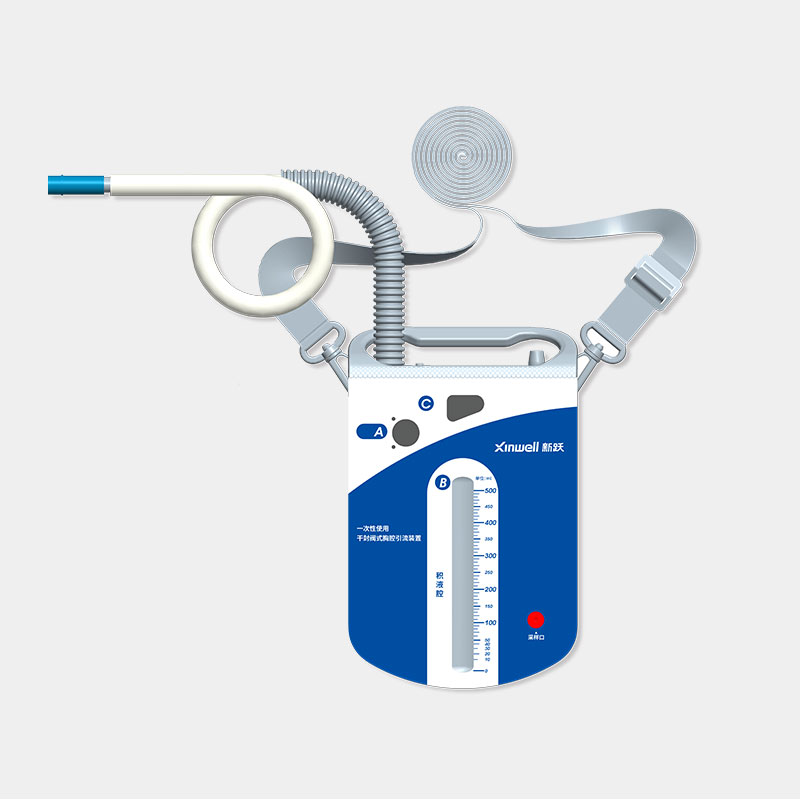
The company has established a quality management system that complies with the requirements of medical device regulation. Products and solutions are promoted and applied in more than 100 countries and regions. Medical device products have obtained NMPA (China), FDA (US), CE (EU), ANVISA (Brazil), PMDA (Japan), KFDA (South Korea) and other medical device certifications, and have established cooperative relations with many world-renowned medical device companies.

Certification
· CE Certificate
· Certificate of FDA Registration
· NMPA Certificate


The company has established a quality management system that complies with the requirements of medical device regulation. Products and solutions are promoted and applied in more than 100 countries and regions. Medical device products have obtained NMPA (China), FDA (US), CE (EU), ANVISA (Brazil), PMDA (Japan), KFDA (South Korea) and other medical device certifications, and have established cooperative relations with many world-renowned medical device companies.

Certification
· CE Certificate
· Certificate of FDA Registration
· NMPA Certificate